

The Macroscopic View
The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); that is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample of the liquid (or solid) in a closed container. Examples:
| substance | vapor pressure at 25oC |
| diethyl ether | 0.7 atm |
| bromine | 0.3 atm |
| ethyl alcohol | 0.08 atm |
| water | 0.03 atm |
The vapor pressure of a liquid varies with its temperature, as the following graph shows for water. The line on the graph shows the boiling temperature for water.
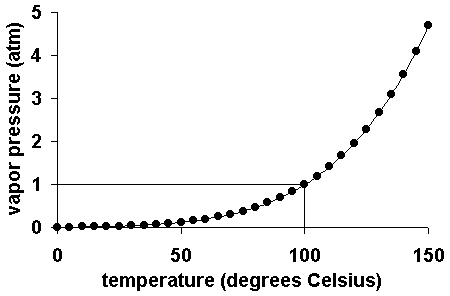
As the temperature of a liquid or solid increases its vapor pressure also increases. Conversely, vapor pressure decreases as the temperature decreases.
The vapor pressure of a liquid can be measured in a variety of ways. A simple measurement involves injecting a little of the liquid into a closed flask connected to a manometer. Click here for an illustration.
The Microscopic View
 |
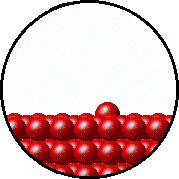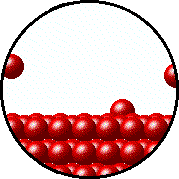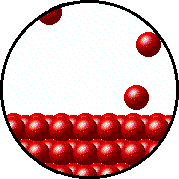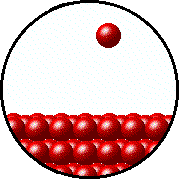 |
| Microscopic equilibrium between gas and liquid. Note that the rate of evaporation of the liquid is equal to the rate of condensation of the gas. | Microscopic equilibrium between gas and solid. Note that the rate of evaporation of the solid is equal to the rate of condensation of the gas. |
|
|
||
| ethyl ether (C4H10O) Pvapor (25oC) = 520 torr The relatively weak dipole-dipole forces and London dispersion forces between molecules results in a much higher vapor pressure compared to ethyl alcohol. |
ethyl alcohol (C2H6O) Pvapor (25oC) = 75 torr Although dipole-dipole forces and London dispersion forces also exist between ethyl alcohol molecules, the strong hydrogen bonding interactions are responsible for the much lower vapor pressure compared to ethyl ether. |
 |
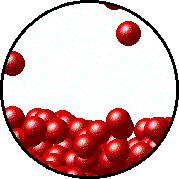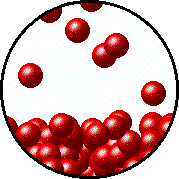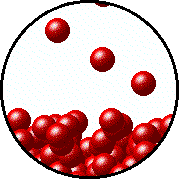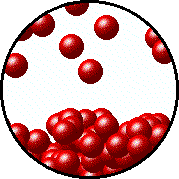 |
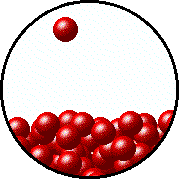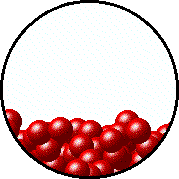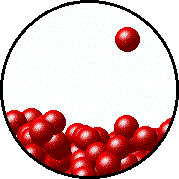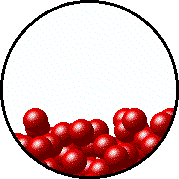
| Microscopic equilibrium between gas and liquid at low temperature. Note the small number of particles in the gas. | Microscopic equilibrium between gas and liquid at high temperature. Note the large number of particles in the gas. |
