Research
The discovery of new catalysts is central to the pursuit of more efficient and sustainable processes in organic synthesis and energy conversion. Research in our group encompasses the various aspects of molecular catalyst design and is broadly aimed at accomplishing fundamentally challenging bond activations and constructions while providing practical solutions to unsolved synthetic problems. Our approach capitalizes on the unique opportunities in the molecular sciences to rationally manipulate catalyst structure in order to control function and probe mechanism. We address the latter in a highly interdisciplinary manner, drawing on a diverse array of physical organic and inorganic, spectroscopic, and computational tools to characterize catalytic intermediates and transition states.
The complex architectures of metalloprotein active sites have been optimized over evolutionary timescales to mediate a range of remarkable chemical reactions. A unifying theme in our group's research is the development of new synthetic platforms that model key aspects of enzyme function without being restricted by the structural motifs available to natural systems. One of our current research interests is elucidating fundamental principles of cooperative substrate activation using multinucleating or multifunctional ligand frameworks. With these design elements as a guiding principle, we are addressing the selective transformation of substrates ranging from small molecules, such as H2 or CO2, to complex organic molecules using earth-abundant, mid-to-late first-row-transition metal centers.
Catalysis at Metal-Metal Bonds
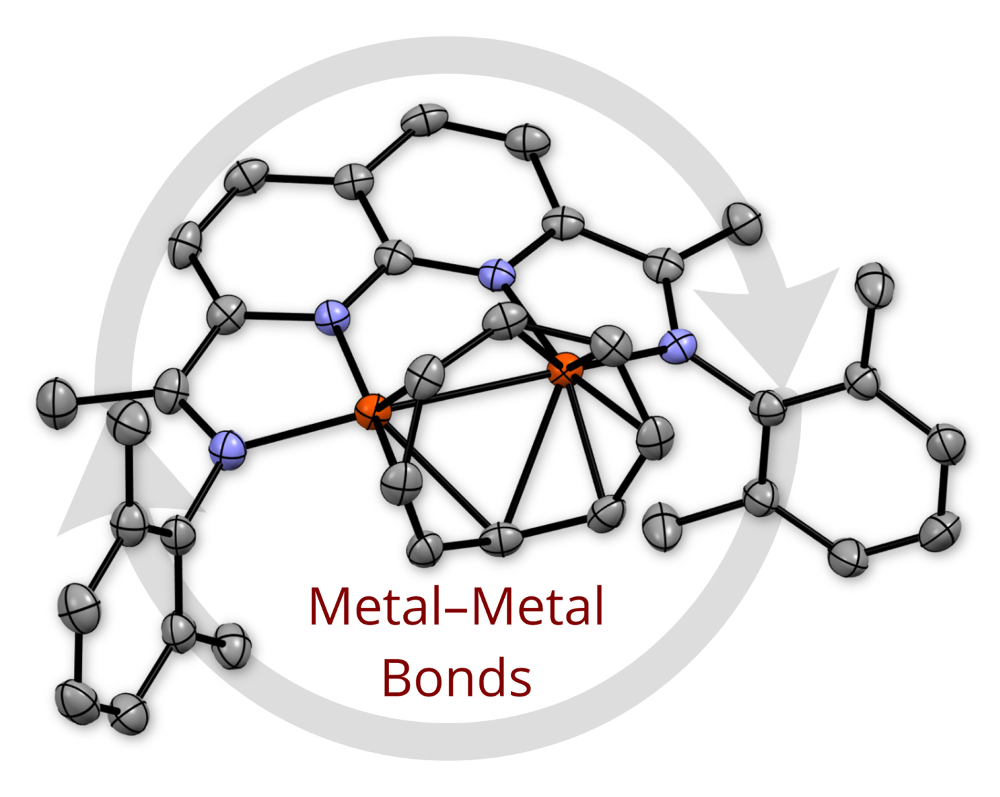 |
An overarching interest of our group is the discovery of new platforms that support coordinatively unsaturated and reactive metal-metal bonds. We have developed a naphthyridine-diimine ligand system that has been used to prepare dinuclear Cu, Ni, Co, and Fe complexes. The redox-active nature of these ligands imparts rich redox chemistry to these complexes, enabling an array of multielectron oxidations and reductions. We are currently exploring the application of these systems to a range of catalytic transformations. |
Organometallic Chemistry
|
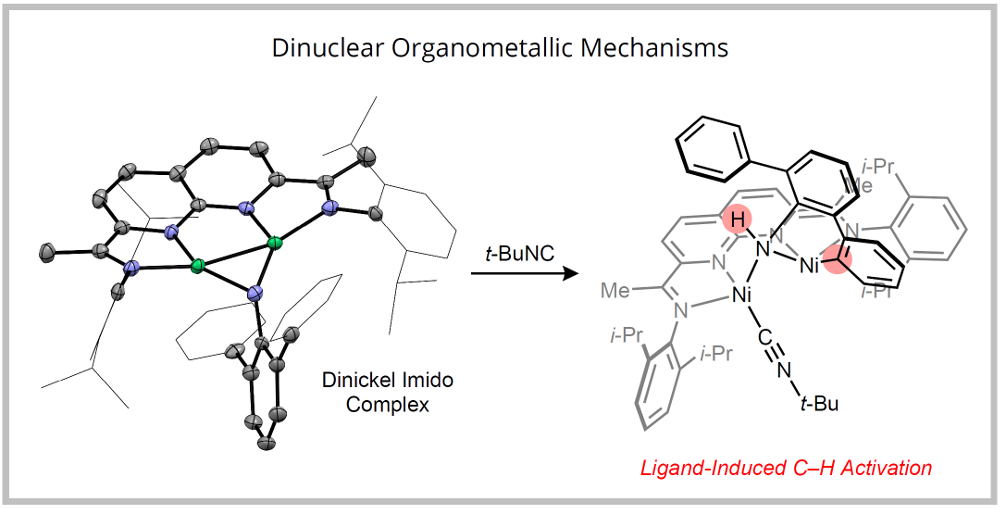
Using the dinuclear platforms developed in our group, we are studying new organometallic mechanisms in which two metals function cooperatively to accomplish bond activation reactions. For example, a well-defined dinickel imido complex undergoes a ligand-induced, C–H activation by a 1,2-addition pathway. We are using these insights to design catalytic processes. |
 |
Reaction Methodology
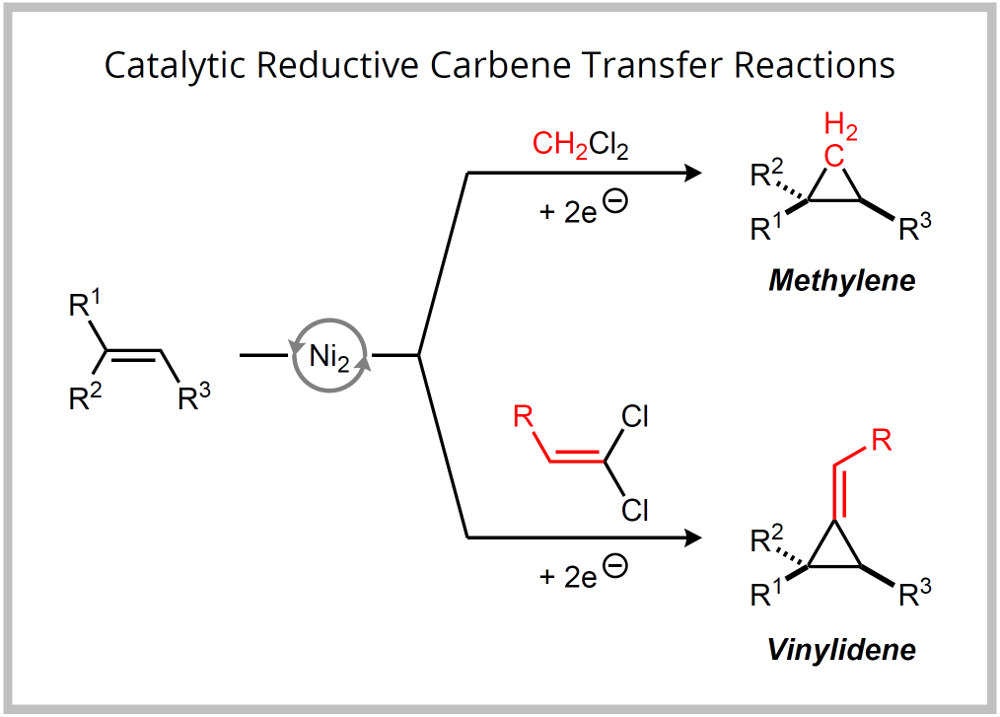 |
A current focus of our methodology program is the development of catalytic reductive carbene transfer reactions. Our approach intercepts carbon–halogen bond activation pathways that have been elucidated in cross-coupling reactions and targets transition metal carbenoid species as key reactive intermediates. We have recently reported catalytic carbene and vinylidene transfer reactions that utilize CH2Cl2 and 1,1-dichloroalkenes as reagents. |
We gratefully acknowledge support from Purdue University, the ACS PRF Doctoral New Investigator Program, the NSF CAREER Program, the NIH MIRA program, and a Sloan Foundation fellowship.
