Field studies
Our research group is actively involved in various field projects focused on the environmental chemistry of the real-world particles. We collect aerosol samples at ground sites and onboard research aircraft and later conduct in-depth laboratory investigations to characterize the size, morphology, phase and composition of collected particles. The qualitative and quantitative information obtained in our studies is essential for evaluating hygroscopic and optical properties of particles, understanding their aging, reactivity, and cloud forming propensity in the atmosphere. Selected examples of our recent Field Environmental Chemistry studies are listed below.
Unrecognized Urban Source of Atmospheric Nanoplastic Particles
Nanoplastic particles are inadequately characterized environmental pollutants that have adverse effects on aquatic and atmospheric systems, causing detrimental effects to human health through inhalation, ingestion and skin penetration. At present, it is explicitly assumed that environmental nanoplastics (EnvNPs) are weathering fragments of microplastic or larger plastic debris that have been discharged into terrestrial and aquatic environments, while atmospheric EnvNPs are attributed solely to aerosolization by wind and other mechanical forces. Our study shows that large quantities of EnvNPs may be directly emitted into the atmosphere as steam-laden waste components discharged from a technology commonly used to repair sewer pipes in urban areas. We suggest that airborne emissions of EnvNPs from these globally used sewer repair practices may be prevalent in highly populated urban areas, and may have important implications for air quality and toxicological levels that need to be mitigated.
(Project supported by NSF grant No. CBET-2107946)
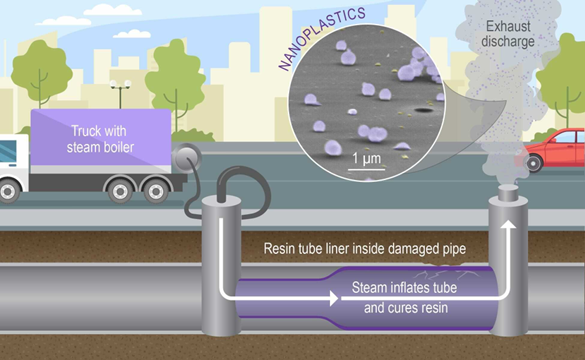
Image Caption: A schematic illustration of cured in place pipe (CIPP) installation. After curing, the newly installed plastic pipe is cooled by blowing forced ambient air through the tube resulting in the atmospheric discharge of waste laden with EnvNP particles.
Relevant publication
A.C. Morales, J.M. Tomlin, C.P. West, F.A. Rivera-Adorno, B.N. Peterson, S.A.L. Sharpe, Y. Noh, S.M.T. Sendesi, B.E. Boor, J.A. Howarter, R.C. Moffet, S. China, B.T. O’Callahan, P.Z. El-Khoury, A.J. Whelton, A. Laskin. Atmospheric emission of nanoplastics from sewer pipe repairs. Nature Nanotechnology, 17, 1171–1177. (2022). doi: 10.1038/s41565-022-01219-9
Molecular Characterization of Water-Soluble Brown Carbon Chromophores in Snowpack Samples
Snowpack is a vital component of the Earth’s climate system because of its high surface albedo providing high reflectivity of solar radiation. Light-absorbing particles deposited on snow significantly reduce the snow albedo and therefore affect the climate through snow albedo feedback. The light-absorbing organic carbon components are termed “brown carbon (BrC)” because of their brown coloring effect on snow. Using advanced molecular-characterization techniques we study various types of water-soluble BrC chromophores in snowpack, including their chemical identifications, relative light absorption contributions, and potential sources, which fill the gap in our knowledge of “unknown” chromophores in snow.
(Project supported by DOE grant No. DE-SC0021977)
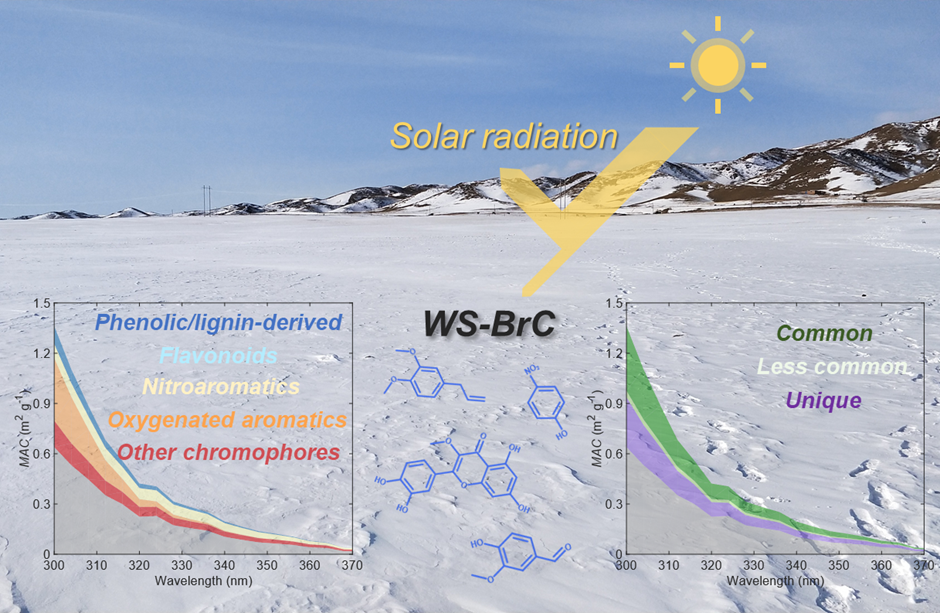
Image Caption: An artwork illustrating contributions of different types of BrC chromophores to the light absorbing properties of snow quantified as mass absorption coefficient (MAC) presented in the insert plots.
Relevant publication
Zhou, C.P. West, A.P.S. Hettiyadura, W. Pu, T. Shi, X. Niu, H. Wen, J. Cui, X. Wang, A. Laskin. Molecular characterization of water-soluble brown carbon chromophores in seasonal snow from northern Xinjiang, China. Environmental Science and Technology, 56 (7), 4173-4186, (2022). doi: 10.1021/acs.est.1c07972
Chemical Composition and Morphological Analysis of Atmospheric Particles from Biomass Burning Emissions
We conduct chemical imaging investigation of individual atmospheric particles collected during biomass burning events. A synchrotron-based scanning transmission X-ray microscopy coupled with near edge X-ray absorption fine structure is employed to probe internally and externally mixed airborne particles and provide quantitative information on their chemical components using a single-particle approach. Description of particle heterogeneity and mixing states based on the chemical imaging results provided quantitative metrics to evaluate transformations of real-world aerosol under intricate environmental conditions.
(Project supported by DOE grant No. DE-SC0018948)
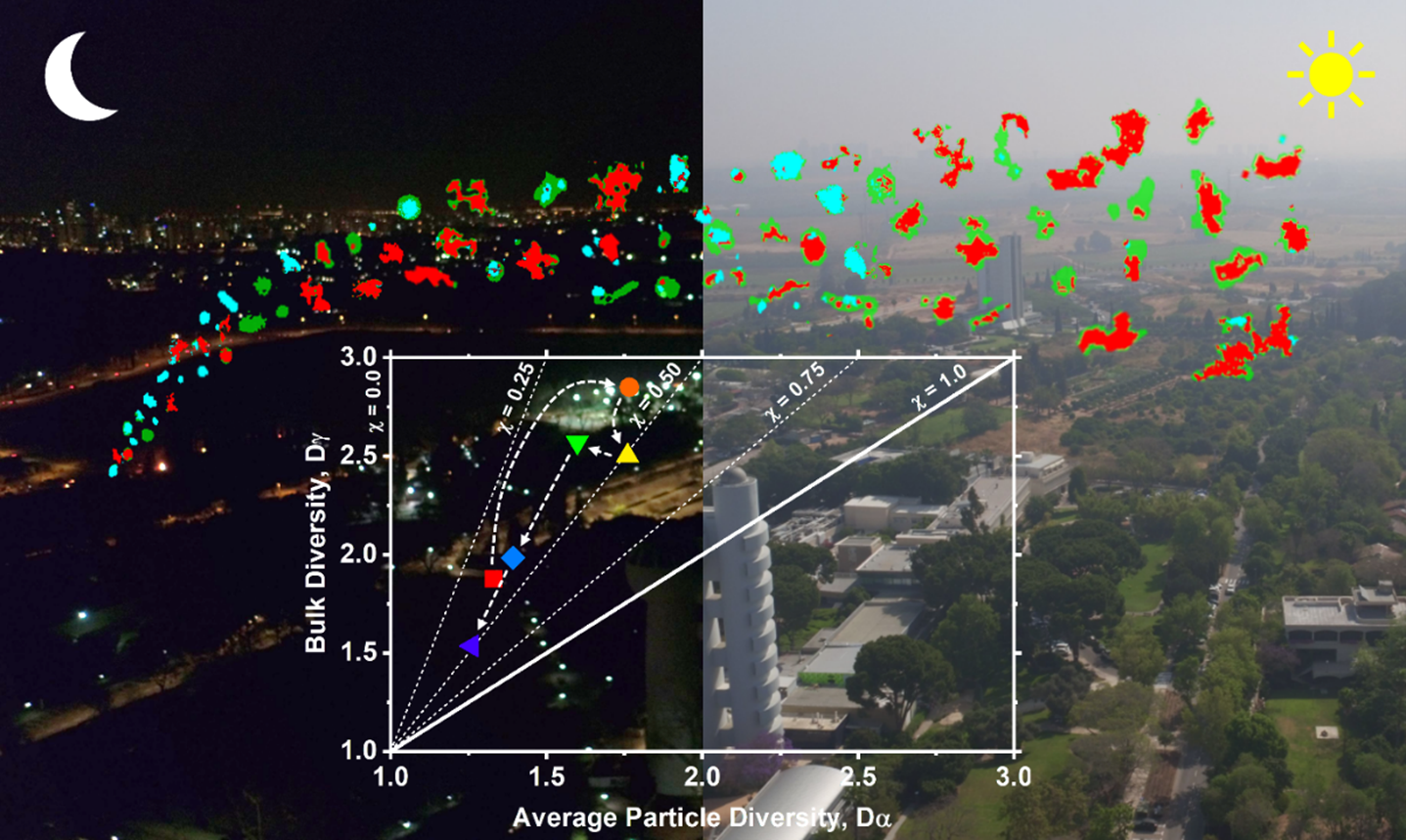
Image Caption: A schematic artwork illustrating characterization of individual particle internal heterogeneity through the perspective of a mixing state framework coupled with the particle-type classification, which provide quantitative metrics to evaluate evolution of complex aerosols under various environmental conditions.
Relevant publication
J.M. Tomlin, J. Weis, D.P. Veghte, S. China, M. Fraund, Q. He, N. Reicher, C. Li, K.A. Jankowski, F.A. Rivera-Adorno, A.C. Morales, Y. Rudich, R.C. Moffet, M.K. Gilles, A. Laskin. Chemical Composition and Morphological Analysis of Atmospheric Particles from an Intensive Bonfire Burning Festival. Environmental Science: Atmospheres, 2, 616–633, (2022). doi: 10.1039/D2EA00037G
Past Projects
