Professor Cooks and team advancing a Green Chemistry catalyst
2020-11-12

Catalysts speed up chemical reactions. And many catalysts contain expensive, ‘noble’ metals. For example, a single load of a catalyst for industrial scale processes (e.g. formation of ethylene oxide from ethylene) can cost as much as $1M. As part of the general “Green Chemistry” movement towards more environmentally friendly, recyclable chemicals, there is growing interest in ‘greener’ catalysts.
Professor R. Graham Cooks and his team are using glass as an inexpensive 'green' catalyst in the Katritzky reaction.
In the current issue of the major chemistry journal, Angewandte Chemie, Yangjie Li, Tsdale F. Mehari, Zhenwei Wei, Yong Liu, and R. Graham Cooks, report that glass particles accelerate an amine transfer reaction by several orders of magnitude. The glass can be recovered and re-used. Initially, they simply washed tiny glass particles from glass wool and saw large reaction rate increases. To demonstrate the effect quantitatively, the authors used glass spheres of 32.5 micron diameter and glass surface (silanol) to reagent ratios of 1:16. They saw a rate increase of 33 times but the reaction did not occur when the silanols on glass were chemically blocked.
Follow-up studies are being done on a variety of reactions. The authors are from Purdue University and Merck & Company (Yong Liu). The work was funded by the National Science Foundation (Grant number CHE-1905087) and the Defense Advanced Research Projects Agency (Award number W911NF-16-2-0020). Purdue University and Merck & Co. have a joint research program in Analytical Chemistry from which this work emerged.
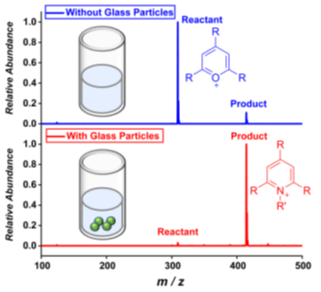
Glass particles accelerate the Katritzky reaction increasing the reaction rate by ca. 100-fold. Evidence shows that glass acts as a ‘green’ heterogeneous catalyst: it participates as a base in the deprotonation step and is recovered unchanged from the reaction mixture.
Citation: DOI: 10.1002/anie.202014613 First Published: 08 November 2020