Chittaranjan Das

- Professor | Biochemistry
- Email: cdas@purdue.edu
- Phone: 45478
- Office: 3131C BRWN
- Professor Chittaranjan Das's individual homepage
The main focus of our lab is to understand the functional role deubiquitinating enzymes (deubiquitinases or DUBs) in cellular pathways, particularly the ones implicated in neurodegeneration (gradual loss of neurons), such as Alzheimer's disease (AD) and Parkinson's disease (PD). A wide variety of biological processes are controlled by the reversible, post-translational modification of proteins by the covalent attachment of ubiquitin, a highly conserved 76-residue eukaryotic polypeptide. Ubiquitination can be thought of as the starting event of a signaling cascade (ubiquitin signaling) that is eventually terminated by the hydrolytic removal of the ubiquitin tag by a DUB. A survey of human genome reveals the presence of about 90 DUBs, suggesting their involvement in a wide variety of biochemical pathways. Our approach to studying DUBs is based on the combined application of a number of tools that include chemical synthesis of small molecule probes, X-ray crystallography, and mass spectrometry-based proteomics.
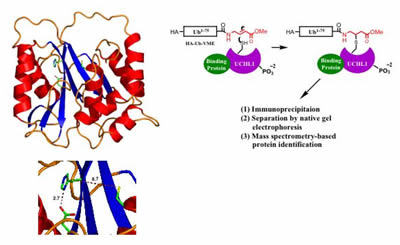
Structure
of UCHL1 determined by X-ray crystallography suggests that the enzyme
can adopt two states, an inactive state (in which the catalytic residues
are misaligned, shown on the left) and a putative active state- in
which the residues are brought into alignment by interaction of the
protein with unknown cofactors. A proposed method for identification of
cofactors of UCHL1 via activity-based purification from whole-cell
extracts (right).
Currently, we are investigating the normal function of the neuronal DUB ubiquitin C-terminal hydrolase L1 (UCHL1)- a PD-associated,neuron-specific protein of unknown physiological function. Our efforts in this direction are aimed at developing cell-permeable small molecule inhibitors of UCHL1 that can be used to probe its function (both normal and disease-associated), determining its binding partners by affinity based purification from whole-cell extracts, and defining the molecular basis of how a naturally occurring variant of this enzyme- in which Ser at the position 18 is substituted by Tyr (called the S18Y polymorph)-provides protection from Parkinson's disease (PD). In addition to UCHL1, we are also conducting structural and mechanistic investigations ofother related enzymes thought to be involved in fundamental biochemical processes such as DNA repair, histone modification, and endocytosis of plasma membrane proteins.
Education
- Ph.D., Indian Institute of Science, 2001
- Postdoctoral Fellow, Harvard Medical School and Brandeis University, 2006
Awards
- Senior Research Fellowship, University Grants Commission, India , 1996