Mark Lipton

- Associate Professor | Organic Chemistry, Chemical Biology
- Email: lipton@purdue.edu
- Phone: 40132
- Office: 210B WTHR
- Professor Mark Lipton's individual homepage
Our research effort combines the disciplines of organic synthesis, bioorganic chemistry and molecular modeling. Current projects in the Lipton group fall into two areas:
Development of novel synthetic methodology.
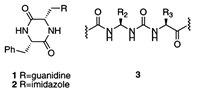
We have recently developed two new reagents for the guanylation of amines, an important and underdeveloped reaction. We also have developed a novel, cyclic dipeptide catalyst ( 1 ) for an asymmetric variant of the Strecker amino acid synthesis. This has led to a broad effort directed toward the use of cyclic dipeptide catalysts (e.g., 2 ) in asymmetric carbon-carbon bond forming reactions. We have initiated another broad effort in the area of reactions on solid supports. We began with the synthesis of cyclic dipeptides 1 and 2 for our catalysis studies. Recent projects involve the synthesis of peptidomimetics (e.g., 3 ) and macrocyclic lactams on solid supports.
Design and synthesis of biologically active molecules.
Ongoing projects include the synthesis of inhibitors of the enzymes cyclophilin A and HIV-1 protease (both essential for the replication of the HIV-1 virus and the pathogenesis of AIDS), and novel DNA-cleaving agents for the treatment of cancer. Projects of this type usually are designed using molecular modeling and tested "in house" after synthesis.
Education
- B.S., Harvey Mudd College, 1981
- M.A., Columbia University, 1984
- M.Ph., Columbia University, 1986
- Ph.D., Columbia University, 1988
- Postdoctoral Fellow, University of California, Berkeley, 1990